In connection with emerging present-day infections, such as HIV/AIDS, SARS, Ebola, and COVID-19, viral sepsis becomes an ever-greater challenge. The elevated cytokine levels and hyper-inflammatory reaction appear as a dominant element in some of the cases. Can an AQP-based therapy be part of the answer to sepsis? This question is most likely to be answered with a personalized approach.
COVID-19 and viral sepsis
COVID-19 and viral sepsis
Although bacterial infections are usually regarded as a leading cause of sepsis, the COVID-19 crisis has revealed viral sepsis to be one of the characteristics of modern-day sepsis syndrome.
Viral sepsis and dysregulated immune reaction
A study of 191 COVID-19 patients treated in December 2019 and January 2020 found that sepsis was the most frequently observed complication. It was diagnosed in 59 percent of all patients included in the study. Furthermore, 100 percent of the non-survivor patients were diagnosed with sepsis. Of the patients diagnosed with sepsis, 76 percent tested negative for bacterial and fungus in their blood and lower respiratory tract specimen cultures. Therefore, these patients can be accurately diagnosed with viral sepsis.118
The elevation of cytokine levels is hypothesized to be a leading cause of the high mortality of COVID-19 and viral sepsis. Cytokines are a group of proteins that control inflammation in our body through a process called cell signaling (communication between cells). When we get an infection, our immune system releases more cytokines. Unfortunately, sometimes the body goes into overdrive and releases too many cytokines. As the body loses control of cytokine production, a cytokine storm is created. Cytokine storms might explain why some people have severe reactions to coronaviruses while others only experience mild symptoms.
Based on the data observed in severe COVID-19 patients in Wuhan, doctors linked cytokine storms to viral sepsis and critically ill COVID-19 patients. Data showed that pro-inflammatory cytokines and chemokines were significantly elevated in COVID-19 patients. It was suggested that the cytokine storm might play an important role in the immunopathology of COVID-19.119
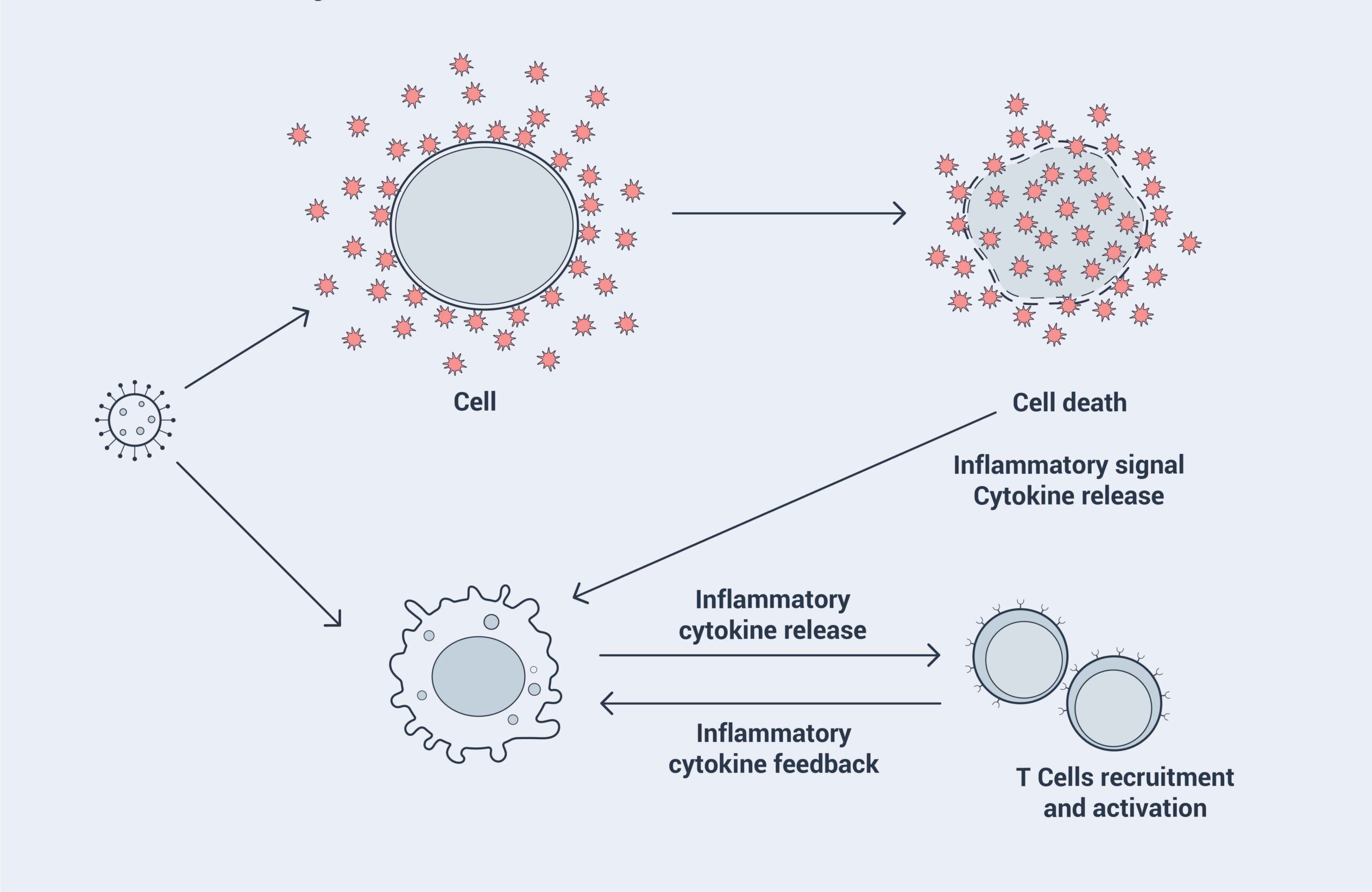
The hypothesis of a cytokine storm was reflected in our interview with Supertrends expert Dr. Masab Moumneh, who has been an ICU physician in Abu Dhabi for the past ten years. Of the 700 beds available in the hospital where Dr. Moumneh works, 110 are ICU beds. “I have never seen anything as bad as this. COVID-19 patients were killed by cytokine storms,” he notes. Dr. Moumneh’s team also observed that cytokine levels can often indicate whether a critically ill patient will survive.
The double-edged sword of immune response
Although it is recognized that cytokine levels were elevated in COVID-19 patients, the precise role of cytokine responses is still unclear. It is also unclear whether these levels cross the threshold from protective to pathological.120 Will the use of immunosuppressants block host immune responses and cause impaired clearance of the virus, thus increasing the risk of secondary infections? None of the studies conducted so far have provided answers.
As for clinicians dealing with COVID-19 patients, the use of immunosuppressants is a difficult choice. Many new drugs designed for treating patients with chronic inflammatory conditions have extensive half-lives, which can cause long-lasting suppression of body’s immune function.121 Furthermore, the complexity of their usage and adverse effects means they are difficult to administer. In the official guideline of sepsis management, no immunomodulators were included.122 Dexamethasone (a type of corticosteroid medication) was the only immunomodulator that was recommended for regulating overreacting immune systems in the guidance on “Care of Critically Ill Patients With COVID-19”, published by US National Institutes of Health.123
Finding new approaches
Based on their observations of elevated cytokine levels, Dr. Moumneh and his colleagues administered corticosteroids to their most severely affected COVID-19 patients, aiming to reduce the overreaction of the immune system. They also attempted to use other immunosuppressants; however, these drugs were not well-tolerated due to severe adverse effects.

Dr. Mads Koch Hansen is another Supertrends expert with vast experience not only in sepsis management, but also in hospital administration. He has worked as clinical lead and medical director for Lillebaelt Hospital in Denmark and was appointed as president and chairman of the Danish Medical Association from 2010 to 2015. Since there is no specific drug for sepsis, he felt that doctors’ hands were quite tied when it came to treating sepsis patients. He welcomes the idea of a new approach to regulate the immune reaction in COVID-19 patients. “From the view of a clinician, the criterial for a successful new approach are that it should be effective, well-tolerated, and easy to administer,” Dr. Hansen told Supertrends.
For many years, scientists have found evidence that AQPs facilitate both the phagocytic functions and the migration of immune cells.124 125 Furthermore, AQPs have been shown to contribute to regulating fluid trafficking and inflammation process in lung infection.126 Swedish start-up Apoglyx has been working on AQP-based therapies since 2012. The pre-clinical results of their AQP-based drug candidate on sepsis were successful. This came at a time of great need.

In an interview with Supertrends, Dr. Michael Rutzler, CEO and founder of Apoglyx, explained why he thinks their AQP9 inhibitor can be a potential treatment for sepsis.
“In sepsis, the dysfunction of the immune system starts to damage vital organs. The global effects also have an impact on blood circulation. Vital organs, including the brain and kidneys, become impaired. This, in turn, causes further damage to other organs. Through our collaboration with other universities, we found strong evidence that the inhibition of AQP9 demonstrated a protective effect from sepsis in rodent models, especially on heart function. We are hoping to get similar results in humans.”

Rutzler has been collaborating with Professor Giuseppe Calamita of Bari University on AQP research for more than ten years. In one of their latest studies, Calamita used an AQP9 gene knockout mice model (based on a genetic technique to create a mice model that lacks AQP9 in its body) to determine the role of AQP9 in sepsis. “We got a very promising result with more than 25 percent of the mice surviving sepsis. The result demonstrated that AQP9 is involved in the pathophysiology and inflammatory responses of sepsis. This is the first time that we studied AQP’s involvement in sepsis in a living animal model. The results proved that modulated AQP might offer a new approach for sepsis management,” Calamita told Supertrends in an interview.
A New Era for Sepsis Management
A New Era for Sepsis Management
Sepsis was one of the first syndromes to be described and named in the history of medicine, but it is even more relevant in the modern era, given the challenges of an increased older population and co-morbidities. A study involving more than 10 million hospitalized adult sepsis patients in the US found that the incidence of sepsis was disproportionately increased in elderly adults, and that age is an independent predictor of mortality. Furthermore, elderly sepsis survivors require skilled nursing or rehabilitation care more frequently after hospitalization.127
Dr. Koch Hansen has worked in medical administration for many years and understand this all too well. “Today, there are more elderly patients and more patients with chronic conditions. Another factor we should pay attention to is that we do more surgeries on elderly people, with less post-surgery staying days in the hospital. This means we will not be able to detect these patients early if they develop sepsis.”

Currently, there is no specific therapy for sepsis management besides infection control, cardiorespiratory resuscitation, and organ support. A lot of effort has been devoted in the past decades to finding a specific adjunctive therapy targeting a dysregulated immune system, especially on immunosuppressive strategies. The results have been disappointing, with most of these studies failing to show any survival benefit.128
One important reason is insufficient appreciation of the heterogeneity of this syndrome. It is currently agreed that septic patients can experience different underlying pathophysiological mechanisms despite similar clinical presentations. The previously proposed pattern of a proinflammatory phase followed by an anti-inflammatory phase in sepsis is not applicable to all patients.
With the advances in genomics and molecular biology, it is now possible to design patient-specific approaches for sepsis treatment. In 2018, The European Society of Clinical Microbiology and Infectious Diseases published a position paper towards precision medicine in sepsis. The current evidence was summarized with the purpose of translating from theory to clinical practice.129 In a separate report on research priorities for sepsis and septic shock by surviving sepsis campaign, the use of targeted/personalized/precision medicine approaches to determine appropriate therapies for certain patients at the right time was recognized as a top priority.130
In sepsis research and clinical trials, one major challenge has been to determine the functional immune status of each patient in order to select the right patient at the right time for the right treatment. Recent data shows that it is possible to categorize patients at the gene transcription level. In 2016, research done by Davenport and colleagues identified transcriptional sepsis response signatures (SRS). Patients characterized by SRS profile with an immunosuppressed phenotype were associated with a much higher mortality rate. The researchers also found that the SRS profile was not related to timing, which challenges the established simplistic view that an initial pro-inflammatory response is followed by an anti-inflammatory response.131
Such information is essential in order to create opportunities for a precision medicine approach. Based on such information, doctors will be able to determine safely which patient should be given an immune inhibition or an immune adjuvant therapy.
Glossary
Heterogeneity: the state or quality of being diverse in nature. In genetics, the production of identical or similar phenotypes by different genetic mechanisms.
References
118. Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., Xiang, J., Wang, Y., Song, B., Gu, X., Guan, L., Wie, Y., Li, H., Wu, X., Xu, J., Tu, S., Zhang, Y., Chen, H., and Cao, B. 2020. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet, 395(10229), 1054–1062. https://doi.org/10.1016/S0140-6736(20)30566-3.
119. Li, H., Liu, L., Zhang, D., Xu, J., Dai, H., Tang, N., Su, X., and Cao, B. 2020. SARS-CoV-2 and viral sepsis: observations and hypotheses. The Lancet, 395(10235), 1517–1520. https://doi.org/10.1016/S0140-6736(20)30920-X.
120. Mangalmurti, N., and Hunter, C. A. 2020. Cytokine Storms: Understanding COVID-19. Immunity, 53(1), 19–25. https://doi.org/10.1016/j.immuni.2020.06.017.
121. Sinha, P., Matthay, M. A., and Calfee, C. S. 2020. Is a “Cytokine Storm” Relevant to COVID-19?. JAMA Internal Medicine, 180(9), 1152–1154. https://doi.org/10.1001/jamainternmed.2020.3313.
122. Rhodes, A., Evans, L. E., Alhazzani, W., Levy, M. M., Antonelli, M., Ferrer, R., Kumar, A., Sevransky, J. E., Sprung, C. L., Nunnally, M. E., Rochwerg, B., Rubenfeld, G. D., Angus, D. C., Annane, D., Beale, R. J., Bellinghan, G. J., Bernard, G. R., Chiche, J. D., Coopersmith, C., De Backer, D. P., … Dellinger, R. P. 2017. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Medicine, 43(3), 304–377. https://doi.org/10.1007/s00134-017-4683-6.
123. NIH. 2020. “COVID-19 Treatment Guidelines.” Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. 9 October. https://www.covid19treatmentguidelines.nih.gov/therapeutic-management/.
124. Zhu, N., Feng, X., He, C., Gao, H., Yang, L., Ma, Q., Guo, L., Qiao, Y., Yang, H., and Ma, T. 2011. Defective macrophage function in aquaporin-3 deficiency. FASEB journal: official publication of the Federation of American Societies for Experimental Biology, 25(12), 4233–4239. https://doi.org/10.1096/fj.11-182808.
125. Loitto, V. M., Forslund, T., Sundqvist, T., Magnusson, K. E., and Gustafsson, M. 2002. Neutrophil leukocyte motility requires directed water influx. Journal of Leukocyte Biology, 71(2), 212–222.
126. Towne, J. E., Harrod, K. S., Krane, C. M., and Menon, A. G. 2000. Decreased expression of aquaporin (AQP)1 and AQP5 in mouse lung after acute viral infection. American journal of respiratory cell and molecular biology, 22(1), 34–44. https://doi.org/10.1165/ajrcmb.22.1.3818.
127. Martin, G. S., Mannino, D. M., and Moss, M. 2006. The effect of age on the development and outcome of adult sepsis. Critical care medicine, 34(1), 15–21. https://doi.org/10.1097/01.ccm.0000194535.82812.ba
128. Vincent J. L. 2016. Individual gene expression and personalised medicine in sepsis. The Lancet. Respiratory medicine, 4(4), 242–243. https://doi.org/10.1016/S2213-2600(16)00068-0
129. Rello, J., van Engelen, T., Alp, E., Calandra, T., Cattoir, V., Kern, W. V., Netea, M. G., Nseir, S., Opal, S. M., van de Veerdonk, F. L., Wilcox, M. H., and Wiersinga, W. J. 2018. Towards precision medicine in sepsis: a position paper from the European Society of Clinical Microbiology and Infectious Diseases. Clinical Microbiology and Infection, 24(12), 1264–1272. https://doi.org/10.1016/j.cmi.2018.03.011.
130. Coopersmith, C. M., De Backer, D., Deutschman, C. S., Ferrer, R., Lat, I., Machado, F. R., Martin, G. S., Martin-Loeches, I., Nunnally, M. E., Antonelli, M., Evans, L. E., Hellman, J., Jog, S., Kesecioglu, J., Levy, M. M., and Rhodes, A. 2018. Surviving sepsis campaign: research priorities for sepsis and septic shock. Intensive care medicine, 44(9), 1400–1426. https://doi.org/10.1007/s00134-018-5175-z
131. Davenport, E. E., Burnham, K. L., Radhakrishnan, J., Humburg, P., Hutton, P., Mills, T. C., Rautanen, A., Gordon, A. C., Garrard, C., Hill, A. V., Hinds, C. J., and Knight, J. C. 2016. Genomic landscape of the individual host response and outcomes in sepsis: a prospective cohort study. The Lancet. Respiratory Medicine, 4(4), 259–271. https://doi.org/10.1016/S2213-2600(16)00046-1