Aquaporins are a family of membrane water channel proteins that osmotically modulate water fluid homeostasis in several tissues; some of them also transport small solutes such as glycerol. The human body expresses 13 AQPs with specific organ, tissue, and cellular localization. The regulation of AQPs takes place either through gating or through controlled transport and integration to specific membrane compartments. AQPs are involved in inflammation, water balance disorders, brain oedema, cancer, and many other health conditions.
Overview
Overview
Regarding their gene and protein structure, AQPs are a family of highly conserved transmembrane proteins. AQPs consist of six alpha-helices which are spanning the cell membrane, and which are connected via five loops A to E (Figure 4.1 A). So-called asparagine-proline-alanine sequences (NPA motifs) in loops B and E are evolutionary highly conserved features. There is evidence that the NPA motifs are functionally important for water permeation. The six helices form a pore structure (Figure 4.1 B), and for most of the AQPs, the known quaternary structure is a homo-tetramer (Fig. 4.1 C), with the exception of AQP11 forming a homodimer. For AQP4 it has been shown that the tetramers can form oligomeric arrays in membranes. The AQP structure can mediate transport of water and other molecules across cell membranes (Figure 4.2).

Depictions of the Secondary (A), Tertiary (B), and Quaternary (C) structure of an AQP channel. Ex = extracellular space, In = intracellular space, N = N-Terminus of protein, C = C-terminus (adapted from Wikipedia).54
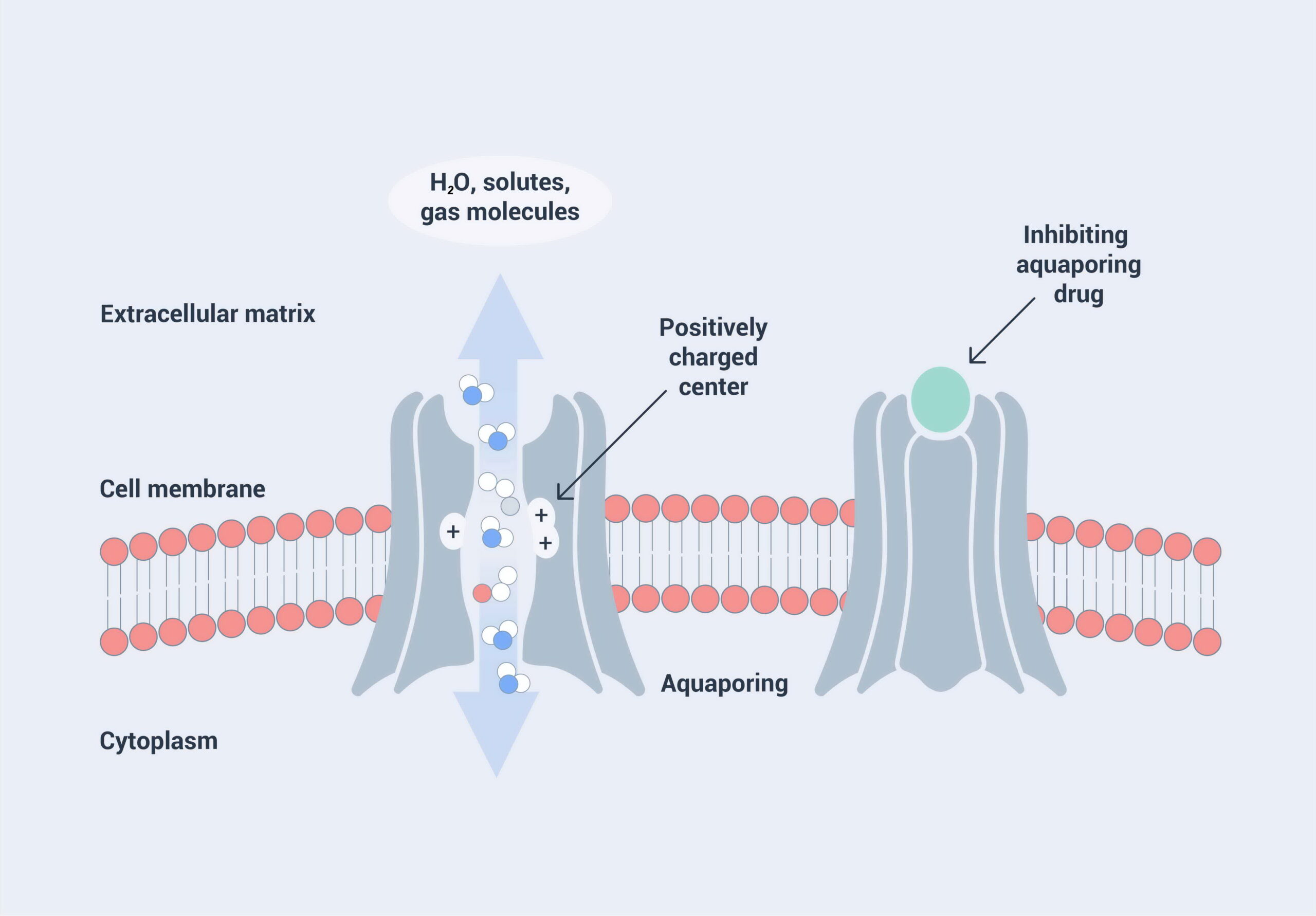
The human body expresses 13 AQPs with specific functions and organ, tissue, and cellular localization as summarized in table 4.1. The regulation of AQPs takes place either through gating or through controlled transport and integration to specific membrane compartments.
Several modulators for AQPs are known. Among these are heavy metal ions (Hg, Pt), some of them antineoplastic drugs (e.g., cisplatin), B-nonylglucoside, tetraethylammonium, thiazolidinediones, sennosides, and auto-antibodies (AQP4) (table 4.1).
| Gene name (Short name; Alias) | Protein function/ location | Location in the human genome (cyto-genetic band) | Protein size, mass, quarternary structure | Known drugs/ inhibitors |
|---|---|---|---|---|
| Major Intrinsic Protein of Lens Fiber (MIP; Aquaporin 0) | Fiber cell membrane protein function is undetermined. May play a role in intracellular communication. Is expressed in the ocular lens and is required for correct lens function. | 12q13.3 | 263 amino acids, 28122 Da, homotetramer. | B-nonylglucoside; Hg2+; Respiratory System Agents |
| Aquaporin 1 (AQP1; Colton Blood Group) | Diseases associated with AQP1 include Blood Group, Colton System and Obstructive Hydrocephalus, aquaporin-mediated transport and O2/CO2 exchange in erythrocytes. | 7p14.3 | Aquaporin 1 (AQP1; Colton Blood Group) | Diseases associated with AQP1 include Blood Group, Colton System and Obstructive Hydrocephalus, aquaporin-mediated transport and O2/CO2 exchange in erythrocytes. |
| Aquaporin 2 (AQP2) | Water channel protein located in the kidney collecting tubule. | 12q13.12 | 271 amino acids, 28837 Da, homotetramer. | Hg2+; thiazolidinediones |
| Aquaporin 3 (AQP3; Gill Blood Group) | Localized at the basal lateral membranes of collecting duct cells in the kidney. Also facilitates the transport of nonionic small solutes such as urea and glycerol, but to a smaller degree. | 9p13.3 | 292 amino acids, 31544 Da. homotetramer. Two isoforms are the molecular basis of the Gill blood group. | Sennosides; Hg2+ |
| Aquaporin 4 (AQP4) | The predominant AQP found in brain and has an important role in brain water homeostasis. | 18q11.2 | 323 amino acids, 34830 Da, Homotetramer. The tetramers can form oligomeric arrays in membranes. | Bevacizumab; Angiogenesis Inhibitors; Autoantibodies; Antineoplastic Agents; AER-271 |
| Aquaporin 5 (AQP5) | Plays a role in the generation of saliva, tears and pulmonary secretions. | 12q13.12 | 265 amino acids, 28292 Da, Homotetramer | Hg2+ |
| Aquaporin 6 (AQP6) | Specific for the kidney. | 12q13.12 | 282 amino acids, 29370 Da. | Hg2+ |
| Aquaporin 7 (AQP1) | Allows movement of water, glycerol and urea across cell membranes. Highly expressed in the adipose tissue where the encoded protein facilitates efflux of glycerol. In the proximal straight tubules of kidney, the encoded protein is localized to the apical membrane and prevents excretion of glycerol into urine. The protein is present in spermatids, as well as in the testicular and epididymal spermatozoa, suggesting an important role in late spermatogenesis. | 9p13.3 | 342 amino acids, 37232 Da, Homotetramer. | Hg2+ |
| Aquaporin 8 (AQP8) | AQP8 mRNA is found in pancreas and colon, but not in other tissues. | 16p12.1 | 261 amino acids, 27381 Da. | Hg2+ |
| Aquaporin 9 (AQP9) | This protein may also facilitate the uptake of glycerol in hepatic tissue and may also play a role in specialized leukocyte functions such as immunological response and bactericidal activity. | 15q21.3 | 295 amino acids, 31431 Da. | Glycerol; Phloretin; Hg2+ |
| Aquaporin 10 (AQP10) | Small intestine AQP; water channel required to promote glycerol permeability and water transport across cell membranes. | 1q21.3 | 301 amino acids, 31763 Da, Homotetramer. | Hg2+ |
| Aquaporin 11 (AQP11) | In several organs like liver, kidney, and brain. | 11q14.1 | 271 amino acids, 30203 Da. Homodimer; disulfide-linked. Can also form homomultimer. | No information |
| Aquaporin 12A (AQP12A) | Expressed in pancreas. | 2q37.3 | 295 amino acids, 31475 Da. | No information |
| Aquaporin 12B (AQP12B) | Expressed in pancreas. | 2q37.3 | 295 amino acids, 31475 Da. | No information |
Loss of Function - Modulation of Function
Loss of Function - Modulation of Function
In this paragraph, we aim to assess how many AQP-based drugs are currently under clinical development for AQP as drug targets. Soon after their discovery, AQPs were considered as potential drug targets.56 57 58 59 60
It is notable that AQPs are of fundamental importance for cellular and body function, and that loss of function would lead to pathological conditions. This can be caused by alterations of a gene, e.g., an AQP gene, which in most cases leads to disruption of the molecular function of the AQP protein. For example, alterations in the Major Intrinsic Protein of Lens Fiber (MIP; Aquaporin 0) gene lead to cataract formation.61 Another way that loss of function can happen is through the formation of autoantibodies. Auto-antibodies are produced by the immune system and are directed against one or more of an individual’s own proteins.
A well-known immune pathological condition arises when autoantibodies are directed against aquaporin 4 (AQP4). This can lead to demyelination and diseases such as optic neuritis or neuromyelitis optica.62
In order to get a more comprehensive idea of which diseases are related to AQP gene or protein loss of function, we assessed a database which serves as an amalgamated human disease compendium with diverse clinical and genetic annotation as well as a structured search information on human diseases from various sources.63 This integrated compendium of annotated diseases was compiled from 68 data sources and holds more than ∼20, 000 disease entries. The results of this research regarding AQPs are listed in table 4.2.
| Gene name (short name) | Disorders |
|---|---|
| Major Intrinsic Protein of Lens Fiber (MIP) | Cataract 15, multiple types; cerulean cataract; early-onset sutural cataract; early-onset posterior polar cataract; early-onset lamellar cataract |
| Aquaporin 1 (Colton Blood Group) (AQP1) | Blood group, Colton system; obstructive hydrocephalus; central pontine myelinolysis; neuromyelitis optica |
| Aquaporin 2 (AQP2) | Diabetes insipidus; nephrogenic, autosomal; diabetes insipidus; diabetes insipidus; neurohypophyseal; syndrome of inappropriate antidiuretic hormone secretion; Ménière’s disease |
| Aquaporin 3 (Gill Blood Group) (AQP3) | Acrokeratoderma; hereditary papulotranslucent; bullous keratopathy; oligohydramnios; Ménière’s disease |
| Aquaporin 4 (AQP4) | Brain oedema; neuromyelitis optica; optic neuritis; myelitis; transverse myelitis |
| Aquaporin 5 (AQP5) | Palmoplantar keratoderma; Bothnian type; aplasia of lacrimal and salivary glands; dry eye syndrome; Sjögren syndrome; Ménière’s disease |
| Aquaporin 6 (AQP6) | Meniere disease; acoustic neuroma; inner ear disease; diabetes insipidus, nephrogenic, autosomal; viral labyrinthitis |
| Aquaporin 7 (AQP7) | Glycerol quantitative trait locus; body mass index quantitative trait locus; fasting hypoglycemia; hydrarthrosis; Ménière’s disease |
| Aquaporin 8 (AQP8) | Polyhydramnios; diabetes insipidus, nephrogenic, autosomal; oligohydramnios; diarrhea |
| Aquaporin 9 (AQP9) | Hydrarthrosis; polyhydramnios; bullous keratopathy; oligohydramnios |
| Aquaporin 10 (AQP10) | Pompholyx; fox-fordyce disease; diabetes insipidus, nephrogenic, autosomal |
| Aquaporin 11 (AQP11) | Diabetes insipidus, nephrogenic, autosomal; failure of tooth eruption, primary |
| Aquaporin 12a (AQP12a) | Inner ear disease; diabetes insipidus, nephrogenic, autosomal |
| Aquaporin 12b (AQP12b) | No data available. |
AQPs: Potential Implications in Inflammation and Other Health Conditions
AQPs: Potential Implications in Inflammation and Other Health Conditions
As a family of membrane water channels, AQPs are involved in a wide range of physiological functions. AQP-mediated osmotic water transport across cell plasma membranes facilitates transepithelial fluid transport, cell migration, and neuroexcitation; AQP-mediated glycerol transport regulates cell proliferation, adipocyte metabolism, and epidermal water retention.65 In general, AQPs play a role in maintaining body water within a suitable range (homeostasis) and in many physiological processes. Through these physiological processes, AQPs affect many health conditions. Inflammation, water balance disorders, brain oedema, and cancer are among the areas that have been extensively studied. There have been very promising developments in connection with small-molecule AQP modulators for clinical applications, including sepsis and cytokine storm, refractory oedema, cancer, glaucoma, certain brain conditions, obesity, etc.66
Aquaporins in inflammation
Inflammation is a multifaceted phenomenon in which the immune system plays a pivotal role. The immune system can be divided into two interconnected subsystems:
- The innate or non-specific immune system includes cells and processes that protect the host from infections by pathogen organisms.
- The adaptive immune response, triggered by the signals from the innate system, releases numerous substances as the real messengers of the inflammation process. The adaptive response, which relies mainly on tissue-resident macrophages and is intermediate between the basal homeostatic state and a classic inflammatory response, is seen as the cause of the chronic inflammatory conditions.67 68
Inflammation is initiated by the alteration of cellular and tissue homeostasis. Cells can undergo modifications of the osmotic microenvironment, causing an increase in the cell’s hydraulic permeability and size.69 Clinically, the swelling of cells and tissues with a surplus of extracellular fluids (oedema) is a clear sign of homeostasis disturbance and inflammation. In some cases, chronic inflammation can be caused by a more intense response and damage.
There is a great deal of data showing that AQPs participate in maintaining constant water homeostasis in many physiological processes related to secretive and absorptive activities.70 AQPs also control cellular processes, including cell adhesion, signaling, volume regulation, and protein expression.71
AQPs and phagocytic functions of immune cells
Cell volume and shape modifications of macrophages promptly occur during phagosome development.72 Aqua-glyceroporins (AQGPs) have been identified as novel key elements in macrophage immune function. Researchers found that AQP3 facilitates water and glycerol transportation and participates in phagocytic and migration activity.73 AQP9 was also confirmed to play a role in macrophages during infection and as a mediator between bacteria and macrophages.74
AQPs and migration of immune cells
Plenty of data has demonstrated that AQPs facilitate migration of immune cells. AQP9 is found to be involved in the migration of neutrophils.75 AQP3 and AQP1 are involved in macrophage migration.76 77
Potential role of AQPs in inflammation of different organs
Researchers have demonstrated that AQPs contribute to regulating fluid trafficking and the inflammation process in lung infection78 and asthma.79 The potential contribution of AQPs in neuroinflammation has also been widely investigated, examining several diseases caused by a failure of innate immunity, such as neuromyelitis optica (NMO) and multiple sclerosis.80 The expression of AQPs and their relevance in physiological and pathological processes have been evidenced in the gastrointestinal tract of human and mammalian species.81 In 2007, Guttman et al. proved the direct correlation between AQPs and diarrhea for the first time, finding that AQP contributes to diarrhea by attaching and effacing bacteria pathogenesis.82 AQPs are also shown to be involved in several inflammatory diseases affecting bone and cartilage. In 2017, Cai et al. identified AQP4 as being potentially responsible for RA pathogenesis in an adjuvant-induced arthritis (AIA) rat model.83
Inflammation is a universal response mechanism that serves as an inter-communicator of biological systems. Uncontrolled or dysregulated inflammation addresses chronic low-grade effects, eventually resulting in multimorbidity. AQPs are clearly potential targets for drugs, paving the way for AQP-based therapies.84
Roles of AQPs in other health conditions
Thirteen AQPs have been identified in mammalian tissues. They are involved in a wide range of physiological functions. With our increasing knowledge of the roles of AQPs in health and disease, AQP-based diagnostics and therapeutics are beginning to be explored. The possibility of regulating AQP expression and function offers potential therapeutic approaches for many pathologies.
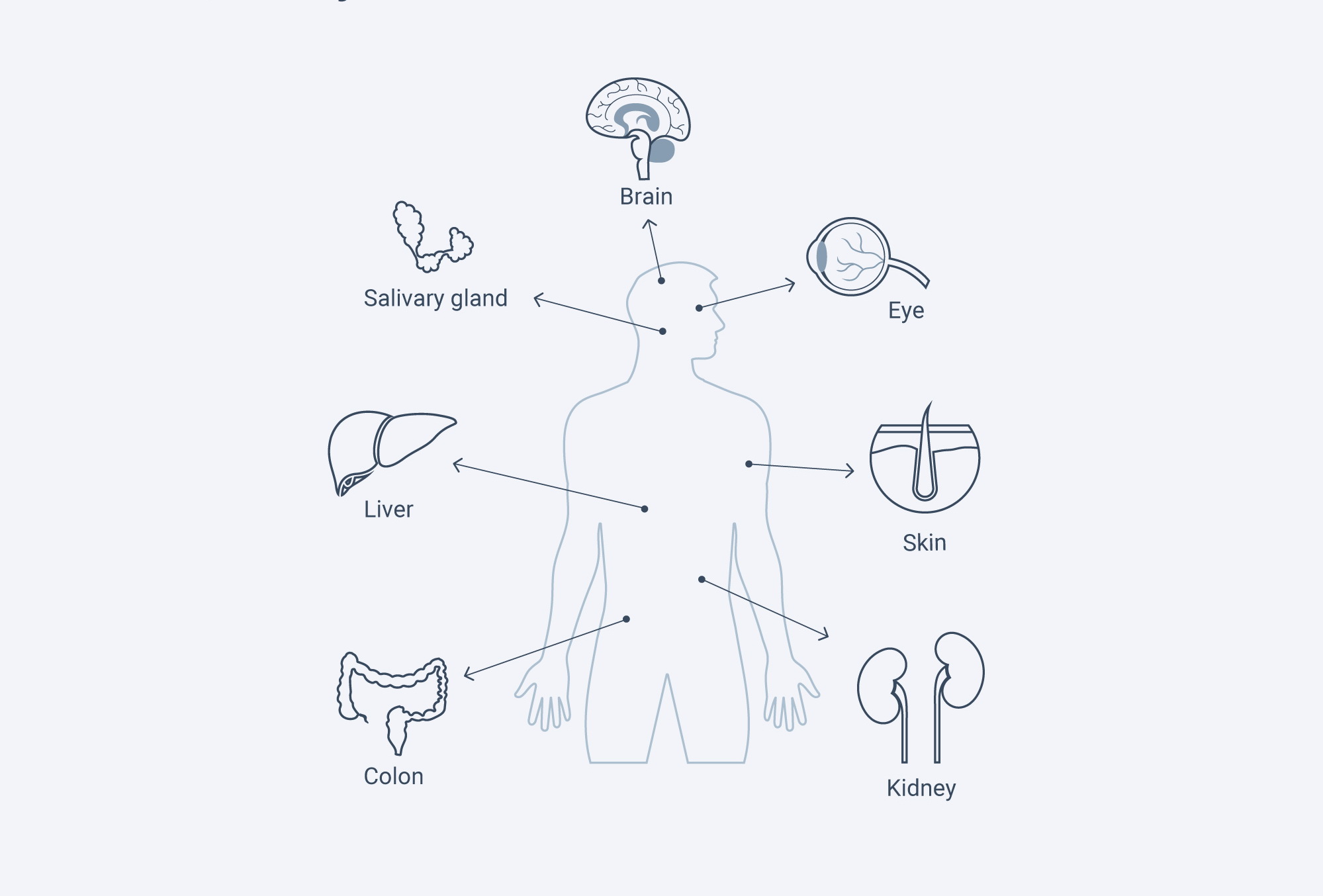
| Location | AQPs | Involvement in health |
|---|---|---|
| Brain | AQP4 | Brain swelling, Epilepsy |
| Epithealia tissues (Salivary gland, pancreas, liver, central nervous system, eye) | AQP1-5, 8 | Glaucoma |
| Blood vessels, other cells | AQP1, 4 | Cancer |
| Kidney | AQP1,2,6 | NDI, Edema |
| Skin | AQP3 | Skin hydration |
| Various cells (such as skin colon, and cornea) | AQP3 | Tumor cell growth, wound healing |
| Adipocytes, liver | AQP3,9 | Obesity, Diabetes |
In kidney and water balance disorders
Kidneys play a key role in body water and electrolyte homeostasis. Among the most closely studied functions of AQPs are their physiological functions in the kidneys. One of the main AQPs expressed in the kidney is AQP1 to 4.85 86 Mutations that cause a lack of AQP1 and AQP2 can result in a urinary concentrating function.87 88 The expression of AQP2 is regulated by vasopressin. Mutations of either the vasopressin or AQP2 gene can lead to congenital nephrogenic diabetes insipidus (NDI). Patients with this condition are unable to concentrate their urine due to lack of AQP2 function, which leads to a high risk of dehydration.89 AQP inhibitors are thus predicted to have “aquaretic” activity, producing a water > salt diuresis. An AQP inhibitor could be a drug (target) for “aquaretic” therapy of refractory oedema.90
In hepatobiliary tract, salivary glands, and pancreas
Recent research has found that AQPs play a role in digestive fluid secretion in the hepatobiliary tract91, salivary glands, and pancreas.92 AQPs are also involved in hepatic gluconeogenesis and fat metabolism in the liver.93 This data indicates that AQPs contribute to the pathogenesis of disorders including cholestasis, gallstone formation, insulin resistance, fatty liver disease, hepatic cirrhosis, and hepatocarcinoma, as well as diabetes.94
In the central nervous system and brain oedema
The association between AQPs, especially AQP4, and brain oedema has been one of the most promising areas for AQPs-based therapy. AQP4 is expressed in astrocytes throughout the Central Nervous System, particularly at the interfaces between brain parenchyma and cerebrospinal fluid in the ventricular and subarachnoid compartments.95
As a bidirectional water channel, AQP4 plays a double role in water accumulation and clearance. In cytotoxic oedema, such as water intoxication, ischemic stroke, and bacterial meningitis, water moves into the brain through an intact blood-brain barrier. AQP4 plays a facilitating role in water accumulation. Underexpression of AQP4 can improve this kind of brain swelling.96 97 Conversely, in vasogenic oedema, such as brain tumors and brain abscesses, water moves into the brain through a leaky blood-brain barrier and exits the brain through AQP4-rich glia limitans. AQP4 plays an important role in water clearance. Underexpression of AQP4 in this model worsens clinical outcome.98 (Papadopoulos et al. 2004). This model also applies to obstructive hydrocephalus.99 Similarly, in spinal cord injury, AQP4 underexpression is associated with reduced swelling in compression injury (cytotoxic oedema), but with increased swelling in contusion injury (vasogenic oedema).100
In cancer
The involvement of AQPs in tumor angiogenesis, local invasion, and metastasis is through AQP-facilitated cell migration.101 Many tumors express AQPs, with the expression correlating with tumor grade.102 Researchers have found impaired tumor growth and angiogenesis in mice lacking AQP1.103 AQP-facilitated cell migration appears to be relevant not only to angiogenesis but also to tumor spread, glial scarring, wound healing, and immune-cell chemotaxis.104
Other than AQPs, AQGP was also found to be involved in cancer. AQGP3 was also found to facilitate cell proliferation in various AQP3-expressing cell types, including skin, colon, and cornea. AQP3-null mice showed complete resistance to formation of skin tumors in response to a tumor initiator-promoter protocol.105 It is possible that AQP3 inhibition can reduce both tumor-cell migration and proliferation. In recent years, various chemicals have been tested as AQP inhibitors for their therapeutic effects on cancer, especially those targeting AQP5 and AQP3.106 Reports also suggest that AQPs may play a role as prognostic markers.107 In the near future, we may see these studies enter a clinical study phase.
Involvement in other conditions
AQPs are involved in many other physiological conditions. Although the mechanisms have not been clearly established, many AQPs provide novel therapeutic approaches for different health conditions.
In neural tissues, AQP4 is expressed in supportive cells adjacent to excitable cells in the brain, retina, inner ear, and olfactory epithelium. Studies have shown impairment in vision, hearing, and olfaction in AQP4-deficient mice.108 (Verkman 2012). Other studies found that AQP4 deficiency reduced the threshold for seizure initiation and increased seizure duration and intensity.109 (Binder et al. 2006).
Studies on AQGP also found that AQP-regulated membrane glycerol permeability is involved in certain physiological functions. AQP3-facilitated glycerol transport in the skin is an important determinant of epidermal and stratum corneum hydration.110 AQP7 is expressed in the plasma membrane of adipocytes, and its deficiency can cause an increase in fat mass and adipocyte hypertrophy.111 AQP9 has also been shown to play an important role in hepatic glycerol uptake.112
Glossary
Angiogenesis: The formation of new blood vessels from existing vessels is called angiogenesis. It is a normal part of growth and healing. It also plays a role in several diseases, including cancer.113
Gluconeogenesis: A process by which our body forms glucose from non-carbohydrate precursors. This process can help to maintain the glucose level when we are not consuming enough carbohydrates.
Macrophage: Macrophage is a type of the blood cells involved in the detection, phagocytosis, and destruction of bacteria and other harmful organisms. They can also activate other immune cells and present antigens to T cells and initiate inflammation by releasing molecules (known as cytokines).114
Neutrophils: Neutrophils constitute another type of blood cell in the immune system. They are one of the first cells that travel to the site of acute inflammation during a bacterial infection. They are part of the innate immune system and the most abundant type of white blood cells in humans. They help to heal damaged tissues and resolve infections, and they form an essential part of the innate immune system. Neutrophils are the first white blood cells recruited to sites of acute inflammation, in response to chemotactic cues produced by stressed tissue cells and tissue-resident immune cells such as macrophages.115
Subarachnoid space: The subarachnoid space is the interval between the arachnoid membrane and the pia mater. CSF pass through the ventricular system and eventually enters the subarachnoid space.116 It is occupied by delicate connective tissue trabeculae and intercommunicating channels containing CSF as well as branches of the arteries and veins of the brain.
Ventriculus of the brain: The ventricles of the brain are a communicating network of cavities filled with cerebrospinal fluid (CSF). They are interconnected and located within the brain parenchyma.117
References
54. Wikipedia 2021. https://en.wikipedia.org/wiki/Aquaporin_4. Accessed January 2021.
55. Stelzer, G., Rosen, N., Plaschkes, I., Zimmerman, S., Twik, M., Fishilevich, S., Stein, T. I., Nudel, R., Lieder, I., Mazor, Y., Kaplan, S., Dahary, D., Warshawsky, D., Guan-Golan, Y., Kohn, A., Rappaport, N., Safran, M., and Lancet, D. 2016. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Current protocols in bioinformatics, 54, 1.30.1–1.30.33. https://doi.org/10.1002/cpbi.5.
56. Beitz, E., and Schultz, J. E. (1999). The mammalian aquaporin water channel family: A promising new drug target. Current medicinal chemistry, 6(6), 457–467.
57. Jeyaseelan, K., Sepramaniam, S., Armugam, A., and Wintour, E. M. 2006. Aquaporins: a promising target for drug development. Expert opinion on therapeutic targets, 10(6), 889–909. https://doi.org/10.1517/14728222.10.6.889.
58. Wang, F., Feng, X. C., Li, Y. M., Yang, H., and Ma, T. H. 2006. Aquaporins as potential drug targets. Acta pharmacologica Sinica, 27(4), 395–401. https://doi.org/10.1111/j.1745-7254.2006.00318.x.
59. Yool A. J. 2007. Functional domains of aquaporin-1: keys to physiology, and targets for drug discovery. Current pharmaceutical design, 13(31), 3212–3221. https://doi.org/10.2174/138161207782341349.
60. Verkman, A. S., Anderson, M. O., and Papadopoulos, M. C. 2014. Aquaporins: important but elusive drug targets. Nature reviews. Drug discovery, 13(4), 259–277. https://doi.org/10.1038/nrd4226.
61. Berry, V., Francis, P., Kaushal, S., Moore, A., and Bhattacharya, S. 2000. Missense mutations in MIP underlie autosomal dominant 'polymorphic' and lamellar cataracts linked to 12q. Nature genetics, 25(1), 15–17. https://doi.org/10.1038/75538.
62. Levy, M., Wildemann, B., Jarius, S., Orellano, B., Sasidharan, S., Weber, M. S., and Stuve, O. 2014. Immunopathogenesis of neuromyelitis optica. Advances in immunology, 121, 213–242. https://doi.org/10.1016/B978-0-12-800100-4.00006-4.
63. Rappaport, N., Twik, M., Plaschkes, I., Nudel, R., Iny Stein, T., Levitt, J., Gershoni, M., Morrey, C. P., Safran, M., and Lancet, D. (2017). MalaCards: an amalgamated human disease compendium with diverse clinical and genetic annotation and structured search. Nucleic acids research, 45(D1), D877–D887. https://doi.org/10.1093/nar/gkw1012.
64. Rappaport, N. et al. 2017
65. Verkman A. S. 2012. Aquaporins in clinical medicine. Annual review of medicine, 63, 303–316. https://doi.org/10.1146/annurev-med-043010-193843.
66. Tradtrantip, L., Jin, B. J., Yao, X., Anderson, M. O., and Verkman, A. S. 2017. Aquaporin-Targeted Therapeutics: State-of-the-Field. Advances in experimental medicine and biology, 969, 239–250. https://doi.org/10.1007/978-94-024-1057-0_16.
67. Takeda, K., Kaisho, T., and Akira, S. 2003. Toll-like receptors. Annual review of immunology, 21, 335–376. https://doi.org/10.1146/annurev.immunol.21.120601.141126
68. Medzhitov R. 2007. Recognition of microorganisms and activation of the immune response. Nature, 449(7164), 819–826. https://doi.org/10.1038/nature06246
69. Maidhof, R., Jacobsen, T., Papatheodorou, A., and Chahine, N. O. 2014. Inflammation induces irreversible biophysical changes in isolated nucleus pulposus cells. PloS one, 9(6), e99621. https://doi.org/10.1371/journal.pone.0099621.
70. Laforenza U. 2012. Water channel proteins in the gastrointestinal tract. Molecular aspects of medicine, 33(5-6), 642–650. https://doi.org/10.1016/j.mam.2012.03.001.
71. Kitchen, P., Day, R. E., Salman, M. M., Conner, M. T., Bill, R. M., and Conner, A. C. (2015). Beyond water homeostasis: Diverse functional roles of mammalian aquaporins. Biochimica et biophysica acta, 1850(12), 2410–2421. https://doi.org/10.1016/j.bbagen.2015.08.023.
72. Clarke, M., Engel, U., Giorgione, J., Müller-Taubenberger, A., Prassler, J., Veltman, D., and Gerisch, G. 2010. Curvature recognition and force generation in phagocytosis. BMC biology, 8, 154. https://doi.org/10.1186/1741-7007-8-154.
73. Zhu, N., Feng, X., He, C., Gao, H., Yang, L., Ma, Q., Guo, L., Qiao, Y., Yang, H., and Ma, T. 2011. Defective macrophage function in aquaporin-3 deficiency. FASEB journal : official publication of the Federation of American Societies for Experimental Biology, 25(12), 4233–4239. https://doi.org/10.1096/fj.11-182808.
74. Holm, A., Karlsson, T., and Vikström, E. 2015. Pseudomonas aeruginosa lasI/rhlI quorum sensing genes promote phagocytosis and aquaporin 9 redistribution to the leading and trailing regions in macrophages. Frontiers in microbiology, 6, 915. https://doi.org/10.3389/fmicb.2015.00915.
75. Loitto, V. M., Forslund, T., Sundqvist, T., Magnusson, K. E., and Gustafsson, M. 2002. Neutrophil leukocyte motility requires directed water influx. Journal of leukocyte biology, 71(2), 212–222.
76. Zhu, N. et al. 2011
77. Tyteca, D., Nishino, T., Debaix, H., Van Der Smissen, P., N'Kuli, F., Hoffmann, D., Cnops, Y., Rabolli, V., van Loo, G., Beyaert, R., Huaux, F., Devuyst, O., and Courtoy, P. J. (2015). Regulation of macrophage motility by the water channel aquaporin-1: crucial role of M0/M2 phenotype switch. PloS one, 10(2), e0117398. https://doi.org/10.1371/journal.pone.0117398.
78. Towne, J. E., Harrod, K. S., Krane, C. M., and Menon, A. G. 2000. Decreased expression of aquaporin (AQP)1 and AQP5 in mouse lung after acute viral infection. American journal of respiratory cell and molecular biology, 22(1), 34–44. https://doi.org/10.1165/ajrcmb.22.1.3818.
79. Krane, C. M., Deng, B., Mutyam, V., McDonald, C. A., Pazdziorko, S., Mason, L., Goldman, S., Kasaian, M., Chaudhary, D., Williams, C., and Ho, M. W. 2009. Altered regulation of aquaporin gene expression in allergen and IL-13-induced mouse models of asthma. Cytokine, 46(1), 111–118. https://doi.org/10.1016/j.cyto.2008.12.018.
80. Oklinski, M. K., Skowronski, M. T., Skowronska, A., Rützler, M., Nørgaard, K., Nieland, J. D., Kwon, T. H., and Nielsen, S. 2016. Aquaporins in the Spinal Cord. International journal of molecular sciences, 17(12), 2050. https://doi.org/10.3390/ijms17122050.
81. Laforenza U. 2012
82. Guttman, J. A., Samji, F. N., Li, Y., Deng, W., Lin, A., and Finlay, B. B. 2007. Aquaporins contribute to diarrhoea caused by attaching and effacing bacterial pathogens. Cellular microbiology, 9(1), 131–141. https://doi.org/10.1111/j.1462-5822.2006.00773.x
83. Cai, L., Lei, C., Li, R., Chen, W. N., Hu, C. M., Chen, X. Y., and Li, C. M. 2017. Overexpression of aquaporin 4 in articular chondrocytes exacerbates the severity of adjuvant-induced arthritis in rats: an in vivo and in vitro study. Journal of inflammation (London, England), 14, 6. https://doi.org/10.1186/s12950-017-0153-8.
84. Mariajoseph-Antony, L. F., Kannan, A., Panneerselvam, A., Loganathan, C., Shankar, E. M., Anbarasu, K., and Prahalathan, C. 2020. Role of Aquaporins in Inflammation – A Scientific Curation. Inflammation, 43(5), 1599–1610. https://doi.org/10.1007/s10753-020-01247-4.
85. Verkman A. S. 2008. Dissecting the roles of aquaporins in renal pathophysiology using transgenic mice. Seminars in nephrology, 28(3), 217–226. https://doi.org/10.1016/j.semnephrol.2008.03.004.
86. Noda, Y., Sohara, E., Ohta, E., and Sasaki, S. 2010. Aquaporins in kidney pathophysiology. Nature reviews. Nephrology, 6(3), 168–178. https://doi.org/10.1038/nrneph.2009.231.
87. King, L. S., Choi, M., Fernandez, P. C., Cartron, J. P., and Agre, P. 2001. Defective urinary concentrating ability due to a complete deficiency of aquaporin-1. The New England journal of medicine, 345(3), 175–179. https://doi.org/10.1056/NEJM200107193450304.
88. Deen, P. M., Verdijk, M. A., Knoers, N. V., Wieringa, B., Monnens, L. A., van Os, C. H., and van Oost, B. A. 1994. Requirement of human renal water channel aquaporin-2 for vasopressin-dependent concentration of urine. Science (New York, N.Y.), 264(5155), 92–95. https://doi.org/10.1126/science.8140421
89. Soveral, G., Nielsen, S., and Casini A. (eds.) 2016. Aquaporins in health and disease. CRC Press. ISBN 978-1-138-89401-3.
90. Verkman A. S. 2012.
91. Boyer J. L. 2013. Bile formation and secretion. Comprehensive Physiology, 3(3), 1035–1078. https://doi.org/10.1002/cphy.c120027.
92. Delporte C. 2014. Aquaporins in salivary glands and pancreas. Biochimica et biophysica acta, 1840(5), 1524–1532. https://doi.org/10.1016/j.bbagen.2013.08.007.
93. Lebeck, J., Cheema, M. U., Skowronski, M. T., Nielsen, S., and Praetorius, J. 2015. Hepatic AQP9 expression in male rats is reduced in response to PPARα agonist treatment. American Journal of Physiology. Gastrointestinal and liver physiology, 308(3), G198–G205. https://doi.org/10.1152/ajpgi.00407.2013.
94. Soveral, G. et al. 2016
95. Verkman A. S. 2012
96. Manley, G. T., Fujimura, M., Ma, T., Noshita, N., Filiz, F., Bollen, A. W., Chan, P., and Verkman, A. S. 2000. Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke. Nature Medicine, 6(2), 159–163. https://doi.org/10.1038/72256.
97. Papadopoulos, M. C., and Verkman, A. S. 2005. Aquaporin-4 gene disruption in mice reduces brain swelling and mortality in pneumococcal meningitis. The Journal of Biological Chemistry, 280(14), 13906–13912. https://doi.org/10.1074/jbc.M413627200.
98. Papadopoulos, M. C., Manley, G. T., Krishna, S., and Verkman, A. S. 2004. Aquaporin-4 facilitates reabsorption of excess fluid in vasogenic brain edema. FASEB journal: official publication of the Federation of American Societies for Experimental Biology, 18(11), 1291–1293. https://doi.org/10.1096/fj.04-1723fje.
99. Bloch, O., Auguste, K. I., Manley, G. T., & Verkman, A. S. 2006. Accelerated progression of kaolin-induced hydrocephalus in aquaporin-4-deficient mice. Journal of cerebral blood flow and metabolism: official journal of the International Society of Cerebral Blood Flow and Metabolism, 26(12), 1527–1537. https://doi.org/10.1038/sj.jcbfm.9600306.
100. Verkman A. S. 2012
101. Verkman A. S. 2012
102. Verkman, A. S., Hara-Chikuma, M., and Papadopoulos, M. C. 2008. Aquaporins – new players in cancer biology. Journal of Molecular Medicine, 86(5), 523–529. https://doi.org/10.1007/s00109-008-0303-9.
103. Saadoun, S., Papadopoulos, M. C., Hara-Chikuma, M., and Verkman, A. S. 2005. Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature, 434(7034), 786–792. https://doi.org/10.1038/nature03460.
104. Verkman A. S. 2012
105. Hara-Chikuma, M., and Verkman, A. S. 2008. Prevention of skin tumorigenesis and impairment of epidermal cell proliferation by targeted aquaporin-3 gene disruption. Molecular and cellular biology, 28(1), 326–332. https://doi.org/10.1128/MCB.01482-07.
106. Soveral, G. et al. 2016
107. Sekine, S., Shimada, Y., Nagata, T., Moriyama, M., Omura, T., Watanabe, T., Hori, R., Yoshioka, I., Okumura, T., Sawada, S., Fukuoka, J., and Tsukada, K. 2012. Prognostic significance of aquaporins in human biliary tract carcinoma. Oncology reports, 27(6), 1741–1747. https://doi.org/10.3892/or.2012.1747.
108. Verkman A. S. 2012
109. Binder, D. K., Yao, X., Zador, Z., Sick, T. J., Verkman, A. S., and Manley, G. T. 2006. Increased seizure duration and slowed potassium kinetics in mice lacking aquaporin-4 water channels. Glia, 53(6), 631–636. https://doi.org/10.1002/glia.20318.
110. Hara-Chikuma, M., et al. 2008
111. Hibuse, T., Maeda, N., Funahashi, T., Yamamoto, K., Nagasawa, A., Mizunoya, W., Kishida, K., Inoue, K., Kuriyama, H., Nakamura, T., Fushiki, T., Kihara, S., and Shimomura, I. 2005. Aquaporin 7 deficiency is associated with development of obesity through activation of adipose glycerol kinase. Proceedings of the National Academy of Sciences of the United States of America, 102(31), 10993–10998. https://doi.org/10.1073/pnas.0503291102.
112. Carbrey, J. M., Gorelick-Feldman, D. A., Kozono, D., Praetorius, J., Nielsen, S., and Agre, P. 2003. Aquaglyceroporin AQP9: solute permeation and metabolic control of expression in liver. Proceedings of the National Academy of Sciences of the United States of America, 100(5), 2945–2950. https://doi.org/10.1073/pnas.0437994100.
113. Wikipedia 2021. https://en.wikipedia.org/wiki/Angiogenesis. Accessed January 2021.
114. Saldana J. I. 2021. https://www.immunology.org/public-information/bitesized-immunology/cells/macrophages. Accessed January 2021.
115. Wikipedia 2021. https://en.wikipedia.org/wiki/Neutrophil. Accessed January 2021.
116. Radiopedia 2021. https://radiopaedia.org/articles/subarachnoid-space. Accessed January 2021.
117. Medscape 2021. https://emedicine.medscape.com/article/1923254-overview. Accessed January 2021.